On Wednesday, Kendrick Cromer, a 12-year-old boy from a Washington suburb, became the first patient in the world to receive commercially approved gene therapy that could potentially cure the disease.
For the estimated 20,000 sickle cell patients in the United States eligible for treatment, the beginning of Kendrick's months-long medical journey may offer hope. But it also points to the challenges patients face in seeking new sickle cell treatments.
For a lucky few like Kendrick, this treatment could give them the life they longed for. As a solemn and shy young man, he learned that everyday activities such as riding a bicycle, going outside on a cold day, and playing soccer can trigger attacks of searing pain. I was reading.
“Sickle cell always takes away my dreams and prevents me from doing everything I want to do,” he said. Now he feels like he has a chance to live a normal life.
Near the end of last year, the Food and Drug Administration granted two companies permission to sell gene therapy to patients with sickle cell disease, a genetic disorder of the red blood cells that causes debilitating pain and other medical problems. An estimated 100,000 people in the United States have sickle cell disease, most of whom are black. People are born with the disease by inheriting the mutated gene for the disease from their parents.
Although the treatment has helped patients in clinical trials, Kendrick is Bluebird Bio's first commercial patient in Somerville, Massachusetts. Another company, Vertex Therapeutics of Boston, declined to say whether it had started treating patients with an approved CRISPR gene-editing-based therapy.
Kendrick's family's health insurance agreed to cover the surgery, and he began treatment at Children's National Hospital in Washington. Wednesday's treatment was just the first step. Doctors will harvest his bone marrow stem cells, which Bluebird will genetically modify in his specialized lab for treatment.
That will take months. But before that begins, Bluebird will need hundreds of millions of stem cells from Kendrick, and if the first harvest (which takes six to eight hours) isn't enough, the company plans to try one or two more times.
If that's not enough, Kendrick will have to spend another month preparing for another stem cell extraction.
This entire process is so complex and time-consuming that Bluebird estimates that it can treat cells for only 85 to 105 patients a year, including sickle cell patients as well as much rarer patients. This includes patients with beta-thalassemia, a common disease. receive similar gene therapy.
The medical center also has the capacity to treat only a limited number of gene therapy patients. Each person requires intensive treatment from a specialist. After the patient's stem cell treatment, the patient will need to stay in the hospital for one month. During this time, patients often become seriously ill due to intensive chemotherapy.
Children's National can only accept about 10 gene therapy patients a year.
“This is a huge effort,” said Dr. David Jacobson, chief of the medical center's blood and bone marrow transplant division.
top of waiting list
Kendrick has been preparing for the stem cell collection for the past week. He has been treated for pain at the hospital for weeks, but on his last visit, even morphine and oxycodone couldn't control the pain. He brought a Snoopy pillowcase and a special Spider-Man blanket that his grandmother gave him. And he had a goal.
“I want to heal,” he said.
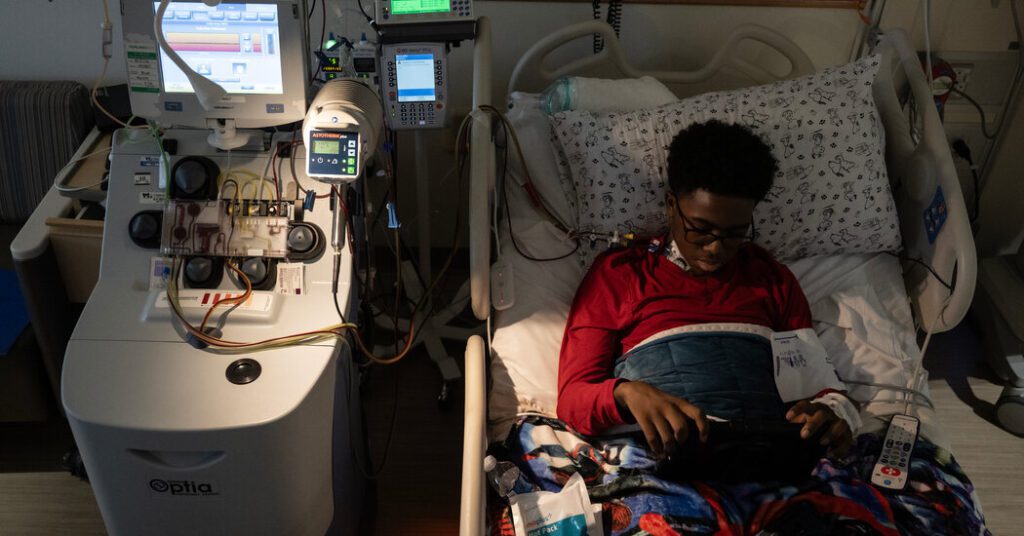
Bone marrow stem cells, the source of all the body's red and white blood cells, are normally found in a person's bone marrow. But Kendrick's doctors injected him with a drug called plerixafor, which forced the blood to float around in his circulatory system.
To isolate the stem cells, hospital staff inserted a catheter into Kendrick's chest vein and attached it to an apheresis machine, a box-like device next to his hospital bed. The blood is spun and separated into plasma, red blood cell, and stem cell layers.
Once enough stem cells are collected, they are sent to Bluebird's lab in Allendale, New Jersey, where technicians add the healthy hemoglobin gene to eliminate the mutated hemoglobin that causes sickle cell disease. Correct the gene.
The corrected cells will be returned to you after 3 months. The goal is to give Kendrick red blood cells that don't form into fragile crescent shapes and get stuck in blood vessels or organs.
Adding new genes to stem cells takes just a few days, but testing for purity, potency, and safety takes weeks to complete. Technicians must grow the cells in the lab before performing these tests.
Bluebird pegs the price of the gene therapy, called Rifgenia, at $3.1 million. This is one of the highest medical costs ever.
Despite the astronomical prices and grueling process, medical centers have waiting lists of patients seeking relief from strokes, organ damage, bone damage, illnesses that can cause severe pain episodes and shortened lifespans. exists.
Children's National has at least 20 patients eligible and interested, Dr. Jacobson said. The choice of who would go first was determined by who was the sickest and whose insurance covered them.
Kendrick qualified on both counts. But while his insurance quickly approved treatment, his insurance pays only a portion of the cost to his family.
chance and hope
Deborah Cromer, a real estate agent, and her husband, Keith, who works in federal law enforcement, had no idea they might have a child with sickle cell.
They first met when Deborah became pregnant with Kendrick. Tests revealed that the baby had a one in four chance of inheriting the mutated gene from her parents and developing sickle cell disease. They may terminate the pregnancy or take a chance.
They decided to take a chance.
The news that Kendrick has sickle cell disease was shocking.
He experienced his first crisis when he was three years old. Sickle cells were trapped in his legs and feet. The baby was inconsolable and Deborah was in so much pain that she couldn't even touch it.
She and Keith took him to Children's National.
“Little did we know that it would lead to multiple visits to the emergency room for us,” Deborah said.
The pain symptoms became more and more severe. It seemed like anything could trigger them, like playing volleyball for 10 minutes or taking a dip in the pool. And when they happened, Kendrick could require five days to a week of hospital treatment to control the pain.
His parents were always with him. Deborah was sleeping on a small bench in her hospital room. Keith was sleeping in his chair.
“We never dreamed of leaving him,” Deborah said.
Eventually, the disease began to take a serious toll. Kendrick developed avascular necrosis, bone death that occurs when blood is lost from the bone, in his lower back. The symptoms also spread to his back and shoulders. He began taking large doses of gabapentin, a nerve pain medication, every day.
His pain never went away. One day he said to Deborah: “Mom, I hurt every day.”
Kendrick wants to be like other children, but fear of pain crisis deters him. He became increasingly sedentary and spent his days using his iPad to watch cartoons and build elaborate Lego structures.
Despite his frequent absences, Kendrick continued to attend school and maintained an A average.
Deborah and Keith began seeking gene therapy. But when they found out how much it would cost, they lost some hope.
But their insurance company approved the treatment within weeks, they said.
Now it has begun.
“We always prayed that this day would come,” Deborah said. But she added: “We have read the agreement and are nervous about what he will have to go through.”
But Kendrick is looking forward to the future. He wants to become a geneticist.
And I said, “I want to play basketball.''