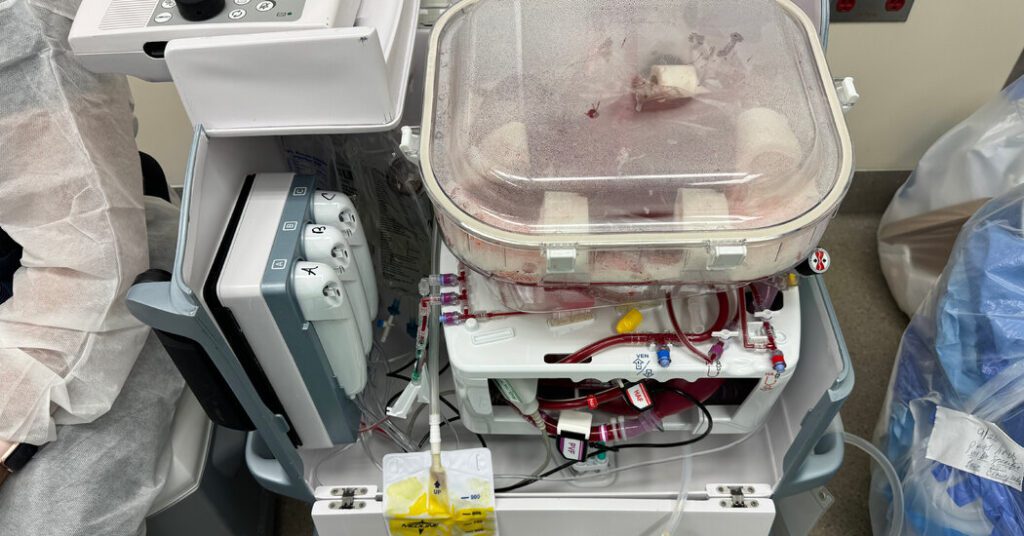
A human liver in an operating room at Chicago's Northwestern Memorial Hospital was alive on some level. Blood circulating through tissues delivers oxygen and removes waste products, and organs produce bile and proteins essential to the body.
However, the donor had died the day before, and the liver was lying in a box-like plastic device. The organ owed its vitality to this machine, which saved the organ for transplantation to a poor patient.
“This is a bit of science fiction,” says Dr. Daniel Borja-Cacho, a transplant surgeon at the hospital.
Surgeons are experimenting with organs from genetically modified animals, hinting at a future in which they could serve as a source for transplants. But the field is already experiencing a paradigm shift with widely used technologies that allow clinicians to temporarily store organs outside the body.
Perfusion, as its name suggests, is changing every aspect of the organ transplant process, from the way surgeons operate, to the types of patients who can donate organs, to recipient outcomes.
Most importantly, more organs are being transplanted with surgical programs that employ perfusion.
Since 2020, the number of liver transplants at Northwestern has increased by 30 percent. Nationally, the number of lung, liver, and heart transplants each increased by more than 10 percent in 2023, one of the largest year-over-year increases in decades.
Without blood flow, organs deteriorate rapidly. For this reason, clinicians have long believed that the ideal organ donor is someone who has died in a situation where brain activity ceases, but whose heart continues to beat, allowing the organ to remain viable until it is matched with a recipient. I've been thinking that there is.
To minimize damage to the organ after removing it from the donor's blood supply and before connecting it to the recipient, surgeons would cool the organ to just below freezing, significantly slowing metabolic processes. .
This extends the period during which organ transplants are available, but only for a short period of time. The liver can survive for less than 12 hours, and the lungs and heart can survive for nearly six hours.
Scientists have long been experimenting with techniques that keep organs in a more dynamic state, at warmer temperatures, and perfused with blood and other oxygen-containing solutions. After years of development, the first device to preserve lungs through perfusion was approved by the Food and Drug Administration in 2019. The device for perfusing the heart and liver was approved in late 2021.
This device essentially pumps blood or oxygenated fluid through a tube into the blood vessels of the donated organ. The cells in the perfused organ continue to function, allowing clinicians to better assess whether the organ will grow within the recipient's body.
Encouraged by that information, transplant surgeons are starting to use organs from older or sicker donors who might otherwise have been turned down, said Dr. Chris Croom, a professor of surgery at the Mayo Clinic in Florida. . “We're going after organs that we would never have gotten before, and we're seeing good results,” he said.
Perfusion also reduces the grueling process of organ recovery and transplantation. These surgeries are hours-long operations that doctors often perform by the hour, starting in the middle of the night and completing in rapid succession.
Surgical teams can now retrieve organs, perfuse them overnight, and complete the transplant in the morning without fear of organ damage caused by delays.
Perhaps most importantly, perfusion further opened the door to organ donations from comatose patients whose families removed life support and eventually allowed their hearts to stop. Tens of thousands of people die each year from such cessation of blood circulation, but because their organs are deprived of oxygen during the death process, they are rarely potential donors.
Currently, surgeons perfuse these organs by removing them into a machine or using less technical methods to recirculate blood to that area of the donor's body. And that made it even more attractive for transplants.
Since 2020, the number of livers transplanted after a donor's cardiovascular death has doubled, according to an analysis of data by the United Network for Organ Sharing, a nonprofit organization that runs the U.S. transplant system.
In the past, surgeons never used hearts from such donors because the organ was sensitive to oxygen deprivation. In 2023, more than 600 transplants were performed thanks to perfusion.
Transplant centers said this new donor cadre will help them more quickly find organs for excess patients in urgent need. Dr. Shimul Shah said the University of Cincinnati's organ transplant program, which he directs, has virtually wiped out the liver waiting list. “I never thought he would say that in his career,” he said.
One barrier to technology adoption can be cost. At the rates currently demanded by device manufacturers, perfusing organs outside the body could increase transplant prices by more than $65,000. Smaller hospitals may not be able to justify the initial cost.
TransMedix, one of the leading companies, received a scathing letter from Arizona Republican Rep. Paul Gosar after it drastically increased prices after regulators approved its device. Ta. is currently being held hostage by a publicly traded company that has lost its true north. ”
However, some surgeons say the technique could save money because patients who receive perfused organs are generally discharged faster, have fewer complications, and have better mid- to long-term outcomes. Ta.
Surgeons are still seeking upper limits on how long perfused organs can survive outside the body, and while the technology is already revolutionizing transplants, this is just the beginning. Some people say
Dr. Shahu Keshavjee, a surgeon at the University of Toronto whose lab has been at the forefront of developing technology to preserve lungs outside the body, said the device would eventually allow doctors to remove lungs rather than replace them. , said it could be possible to repair it and give it back to sick patients. “I think we can create organs that will outlive the person who receives them,” he says.
Dr. Ashish Shah, chief of cardiac surgery at Vanderbilt University, which has one of the busiest heart transplant programs in the country, agreed, calling it the “holy grail.”
“Your heart is the worst,” he said. “I'll take it out. I attached it to my equipment. While you don't have a heart, I'll support you with an artificial heart for a while. Then I'll take your heart out and do cell, mitochondrial, and gene therapy. , whatever else, repair it and then sew your own heart back in. That's what we're really working on.”