The Food and Drug Administration has given the green light to develop new drugs to protect some of the people most at risk from the coronavirus.
The agency has granted emergency use authorization for the monoclonal antibody intravenous drug Pemgarda for immunocompromised patients aged 12 and older. The drug is intended to protect people who are less likely to develop an adequate immune response after vaccination from the new coronavirus. This includes people who have received stem cell or organ transplants and cancer patients who take drugs that suppress the immune system.
“It's going to be for a small percentage of Americans,” said Dr. Michael Mina, a former Harvard University epidemiologist and current chief scientific officer at telemedicine company eMed. But he said those who feel most marginalized at this stage of the pandemic are an important group to protect.
“This is a really important advance for people who are immunocompromised and are literally vulnerable because their immune systems aren't really working,” said Roberts, head of research and development at the Veterans Affairs St. Louis Health System. said one Dr. Ziyad Al Ali. .
David Herring, chief executive officer of Invivid, which makes Pemgarda, said Pemgarda will be available within the next week or two. Herring said the company is still evaluating the drug's price. He expects Medicare and private insurance plans to cover it.
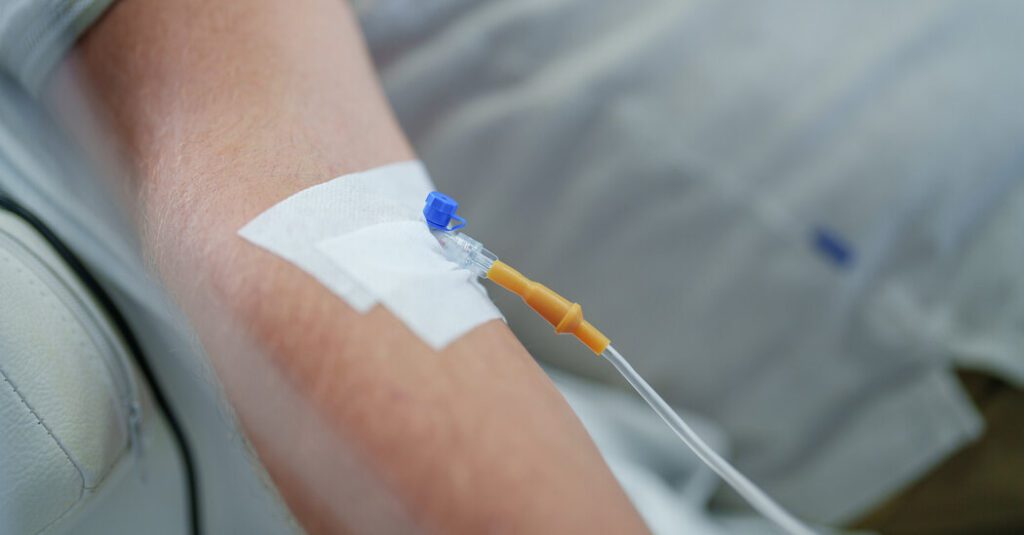
This medication is given as an intravenous drip in a doctor's office or other medical setting. The infusion will take approximately 1 hour to complete. The most common side effects in clinical trials included injection site reactions, cold- or flu-like illness, fatigue, headache, and nausea. Four of the 623 participants in the trial experienced anaphylaxis, a severe allergic reaction.
The goal is to give people drugs before they encounter the virus, reducing their risk of severe outcomes from the coronavirus and reducing their chances of contracting the virus at all. “You can think of it like a vaccine,” Dr. Al-Aly explained. This medicine is not a treatment for the new coronavirus infection. You cannot take Pemgarda if you currently have a virus or have recently been infected.
People may choose to take the drug every three months, Herring said. Dr. Joseph Bailey, a pulmonologist at Northwestern Medicine's COVID-19 Center, said it's unclear whether some people will need additional doses of the drug indefinitely.
Pemgarda is not yet fully approved. “They haven't crossed the research finish line yet,” Dr. Bailey said. The agency approved the drug based on data measuring people's antibody titers, so it's too early to assess exactly how effective it is at preventing infection in the real world.
Previous monoclonal antibody treatments like Evsheld were removed from the market as they became less effective against new coronavirus variants. Herring said the company that makes Pemgarda is preparing to target new variants as they emerge. But Dr. Peter Chin-Hong, an infectious disease expert at the University of California, San Francisco, said it remains to be seen what will actually happen if the virus evolves.
Still, experts are hopeful. “As long as there are still people out there who are extremely vulnerable, the entire community needs to be seriously aware of the virus,” Dr. Mina said.